News
IBDM is recruiting 9 PhD students
Launch your research career with a doctoral contract from Aix-Marseille University through ED62 or through NeuroSchool.
- All
- Awards
- Grant
- Join us
- News
- Research
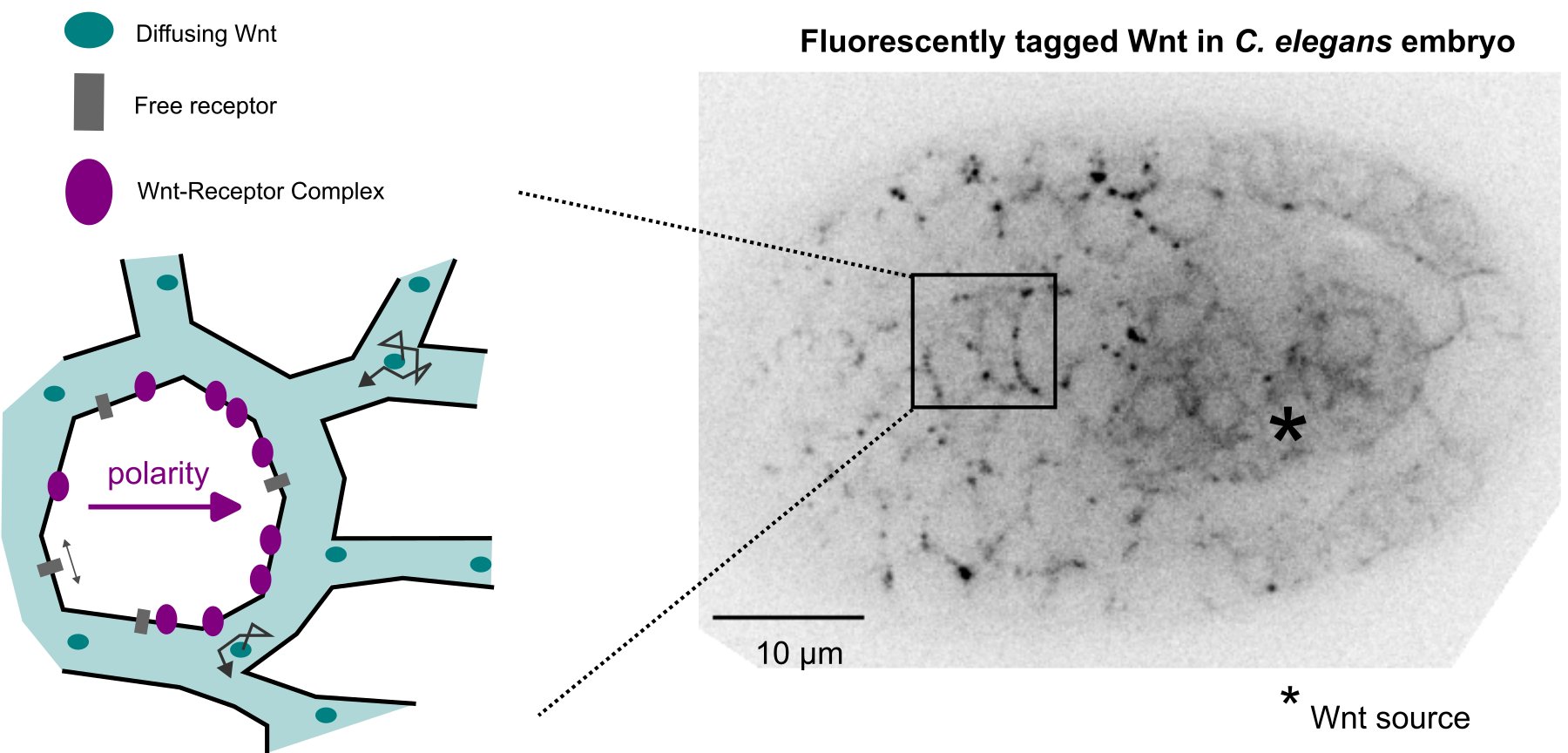
And yet it diffuses!
Free diffusion of Wnt ligands can polarize target cells at distance in C. elegans embryos.
Lost in translation: Vangl2 short and long
Translation starts with a Methionine: True, but not always, as revealed in a study of the PCP component Vangl2.
Emma Legait at the 5th edition of the Neurostories
The brain: a permanent work in progress

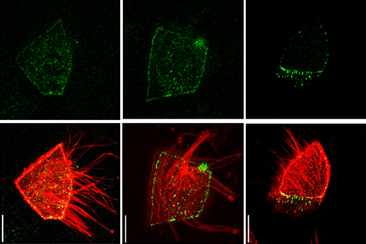
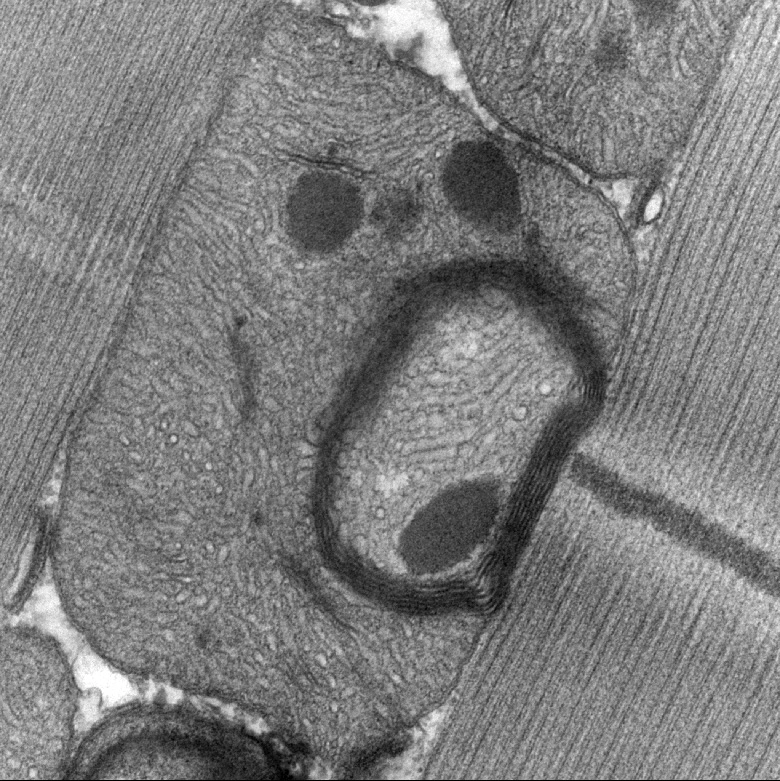
Redefining epithelial junctions with nanoscopy
Lenne and Le Bivic teams show that intestinal adherens junctions are very different from the textbook picture.
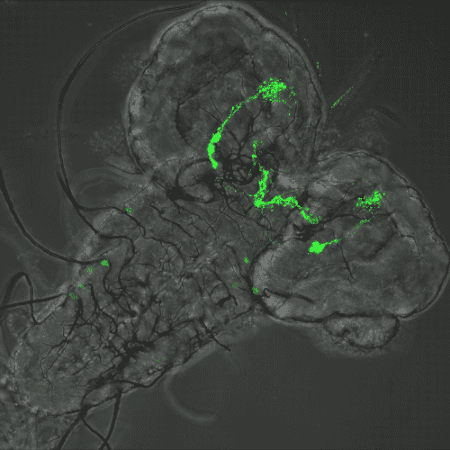
When microbiota shape the host gustatory properties
How the larval microbiota modulates the sensory modalities of future adults?
Zooming in on a wave of tissue invagination
Understanding how adhesion and contractility drive a wave of tissue invagination from a subcellular perspective.
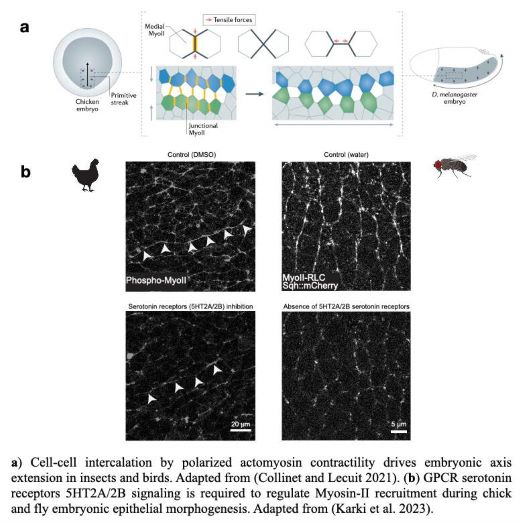
Serotonin on Mechanics and Morphogenesis Mood
Sanjay Karki from Thomas Lecuit’s team reports that serotonin signaling regulates actomyosin contractility during embryonic axis morphogenesis in insect and bird.
Tiny Avengers of Science Uncovering Stem Cell Secrets
Watch how model organisms are used at the IBDM to investigate normal and derailed stem cell trajectories.

The Evolution of an Agricultural Pest: A Sweet Shift
Sweet Temptations: How Drosophila Suzukii’s Love for Sugar is Causing Havoc in the Agriculture Industry.

IBDM welcomes a new research group!
We are delighted to welcome Leo Guignard and his new research group ‘Computer Science, Morphogenesis and Variability’ to IBDM.

Recent achievements and promotions
Celebrating the accomplishments of IBDM members.

Join the IBDM for your internship!
Seeking for your Master internship? The IBDM seems like the right place to do it? Check out our offers.

Youth & Science: Delta Festival
Delta Festival 2023: Music, themed villages, and a touch of IBDM to shine under the Marseille sun.

IBDM teams win 4 grants from ANR, INCa and Fondation ARC
Congratulations to Robert Kelly, Frank Schnorrer, Cédric Maurange, Bianca Habermann and Delphine Delacour!

Alcohol and brain development
The Mann team identifies the microRNA-17-5p as responsible for alcohol-related brain connectivity defects.
Congratulation to our young and talented doctors!
3 motivated and talented students successfully defended their thesis between February and July 2023.

IBDM welcomes a new research group!
We are delighted to welcome Delphine Delacour and her new research group ‘Epithelial monolayer morphogenesis’ to IBDM.

A new model for cell division orientation
The Delacour group proposes a new mechanism for the control of planar spindle orientation and monolayered tissue architecture in the intestinal epithelium.
IBDM gets schoolchildren excited about science!
IBDM Marseille inspires young minds: engaging primary school children on childhood cancer (“Contre le cancer, j’apporte ma pierre”) and interacting with high school students through immersive experiences (DECLICS).

Transcriptional regulation of cellular respiration
The Saurin/Graba group discover the transcription factor M1BP as new major regulator of oxidative metabolism.
Embark on a PhD journey at the IBDM
Launch your research career with a doctoral contract from Aix-Marseille University through ED62 or through NeuroSchool.

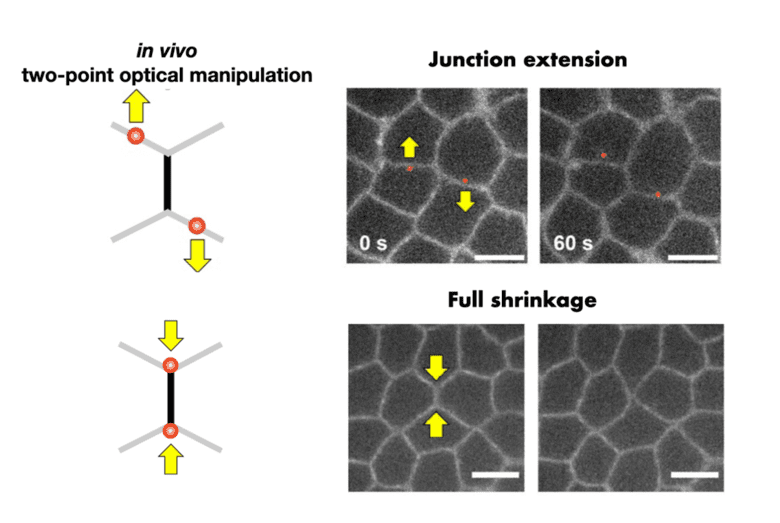
Optical manipulation reveals cell junction mechanics
Epithelial tissues under tension: a study explores how individual cells deform and respond to forces.
Looking back at recent recruitments and promotions
Congratulations to Ruth Souza, Elise Arbeille, Fabien Durbesson, Vincent Bertrand, Léopold Kurz, Olivier Zugasti, Nathalie Caruso, Cédric Matthews, Florian Roguet, Fabienne Lamballe.

Congratulation to our young and talented doctors!
5 motivated and talented students successfully defended their thesis between September 2022 and January 2023.

An ERC Synergy Grant for the BREAKDANCE project
Lenne group, together with 3 other groups, Merkel (CNRS), Trivedi (EMBL), and Ruprecht (CRG) – embark on BREAKDANCE

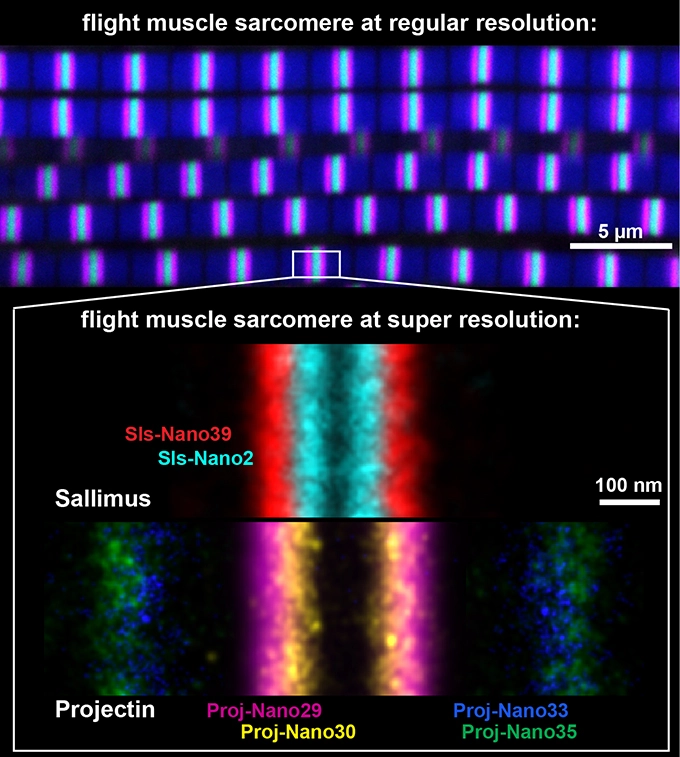
PAINTing muscle with nanobodies positions protein giants
Drosophila sarcomeres visualised at super-resolution with the help of nanobodies and single molecule blinks.
How disruption drives change
Interaction between tight junction formation and gastrulation signals regulates mesendoderm differentiation in hiPSCs
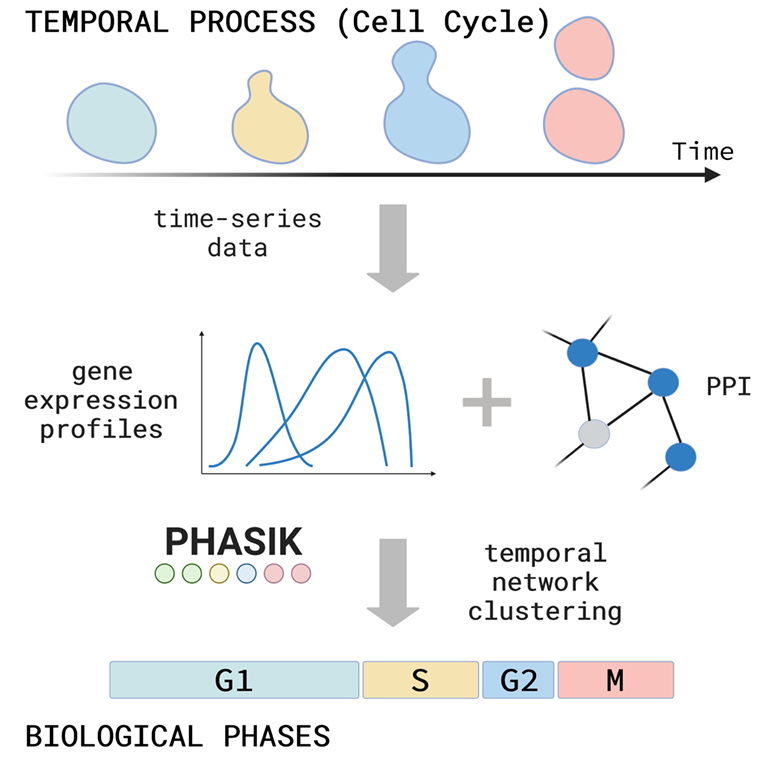
Show me your rhythm!
We introduce an algorithm, Phasik, for extracting the phases of biological systems by clustering partial temporal networks.
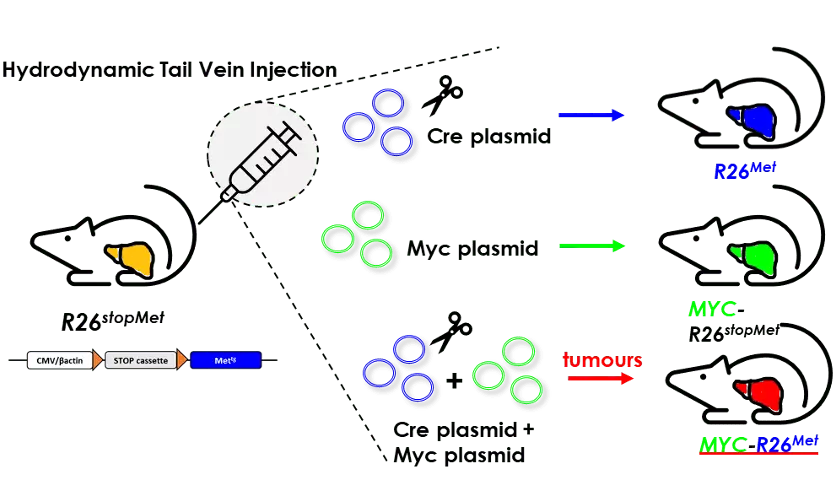
MYC and MET, the ‘Bonnie and Clyde’ in the liver
A new genetic cooperation acting on liver cancer has been uncovered in patients and functionally documented in vivo
Several awards for IBDM members !
Congrats to Aziz Moqrich, Sophie Chauvet and Stefan Harmansa.

The Marseille Developmental Biology Institute is recruiting group leaders
The IBDM invites applications for group leader positions. We seek researchers
who define and address fundamental questions in biology, including the
development, the function, and the dynamics of complex biological systems.
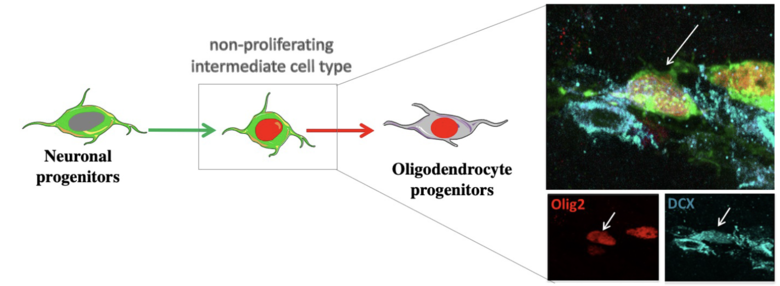
Transformation of neuroblasts into oligodendrocytes
Neuroblasts can generate myelinating oligodendrocytes in pathological states.
Visit of Thierry Lhermitte to the IBDM
Aziz Moqrich team hosted the actor and sponsor of the medical Research Foundation FRM, Thierry Lhermitte.

Organizing the organizers
A new perspective to tackle the very complex cell polarity field.
NeuroStories 2022: Emprise de tête
Mike Altounian presented his work on the effects of alcohol on neural circuit development at Neurostories 2022.

Robert G. Kelly, recipient of the Lamonica Grand Prize for Cardiology
Robert G. Kelly is the recipient of the Lamonica Grand Prize for Cardiology – Fondation pour la Recherche Biomédicale PCL 2022 from the French Academy of Sciences.

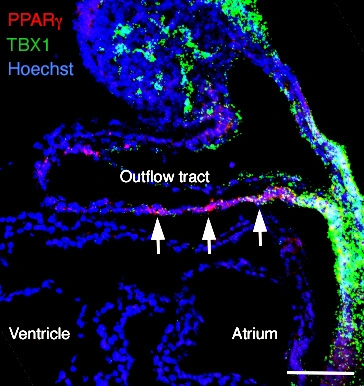
Establishing regional identity in the cardiac outflow tract
Complementary PPARγ and NOTCH pathways pattern outflow tract myocardium
10 IBDM projects have been selected for funding by the ANR
Congratulations to Pascale Durberc, Lucile Miquerol, Aziz Moqrich, Thomas Lecuit, Benjamin Prud’homme, Laurent Fasano, Laurent Kodjabachian, Robert Kelly, Camille Boutin and Sham Tlili.

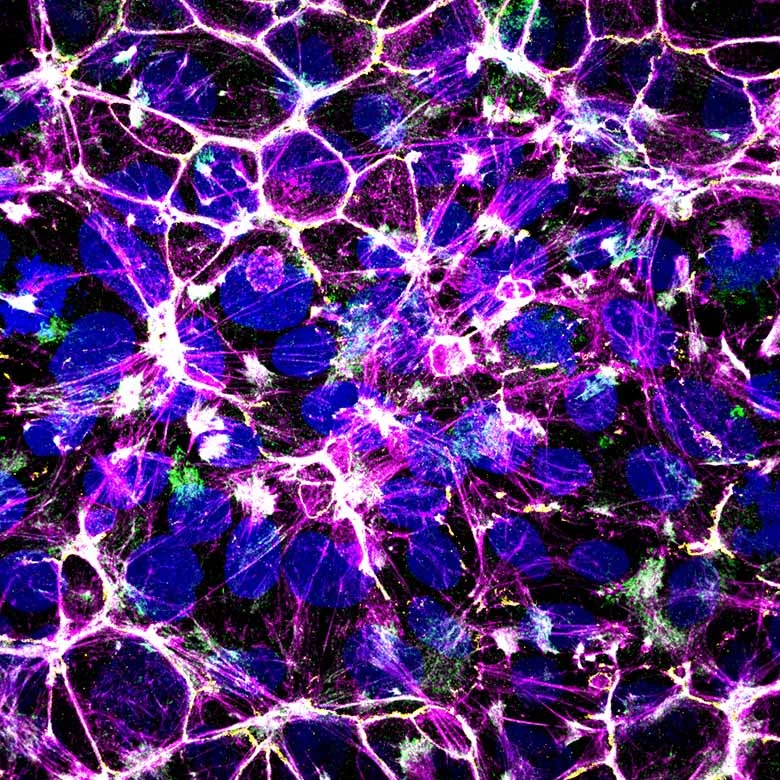
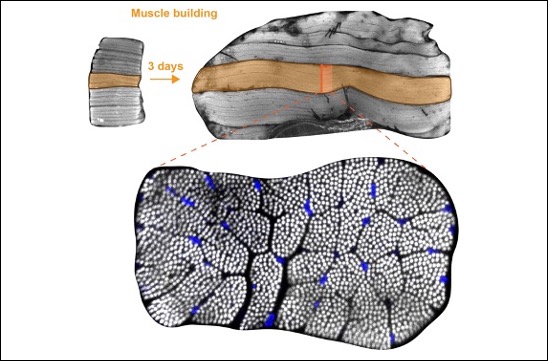
Self-organisation of human muscles in a dish
Human muscle cells self-organise into defined fiber bundles in vitro even without the presence of external cues !

Epigenetic factors regulate tumor hierarchy
PRC2 and MLL1/2 cooperate to guide developmental programs recapitulated in tumors.
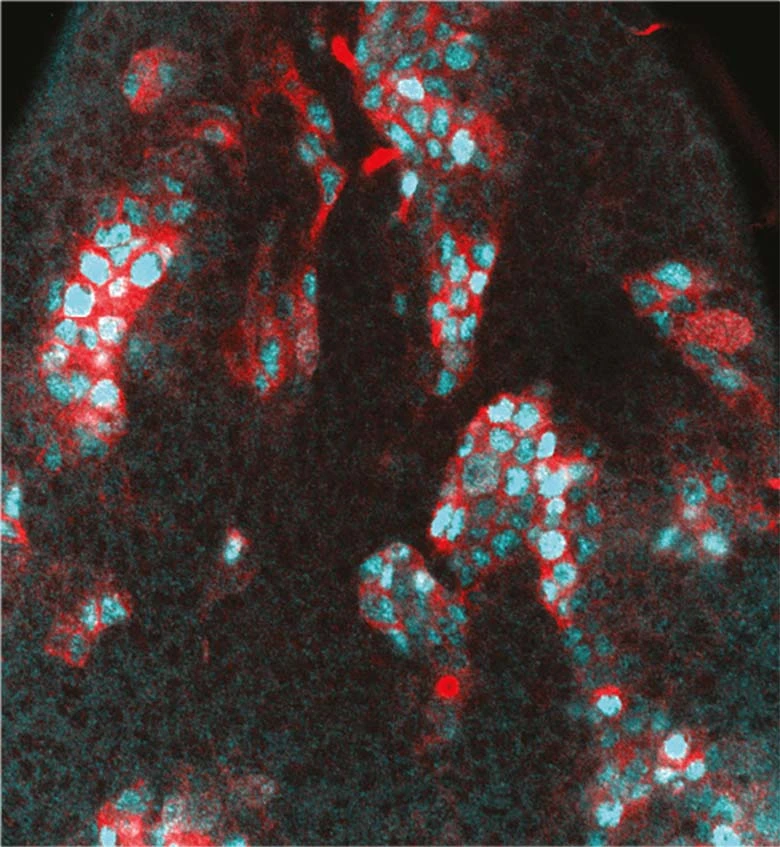
PRC1 chromatin factors ensure the robustness of neuronal differentiation
The Bertrand team shows that PRC1 chromatin factors play a role in the robustness of neuronal fate against gene expression noise and environmental perturbations.
Frank Schnorrer elected as EMBO member
EMBO elects 67 new members and associate members. They join the community of more than 1,900 leading life scientists in Europe and beyond.

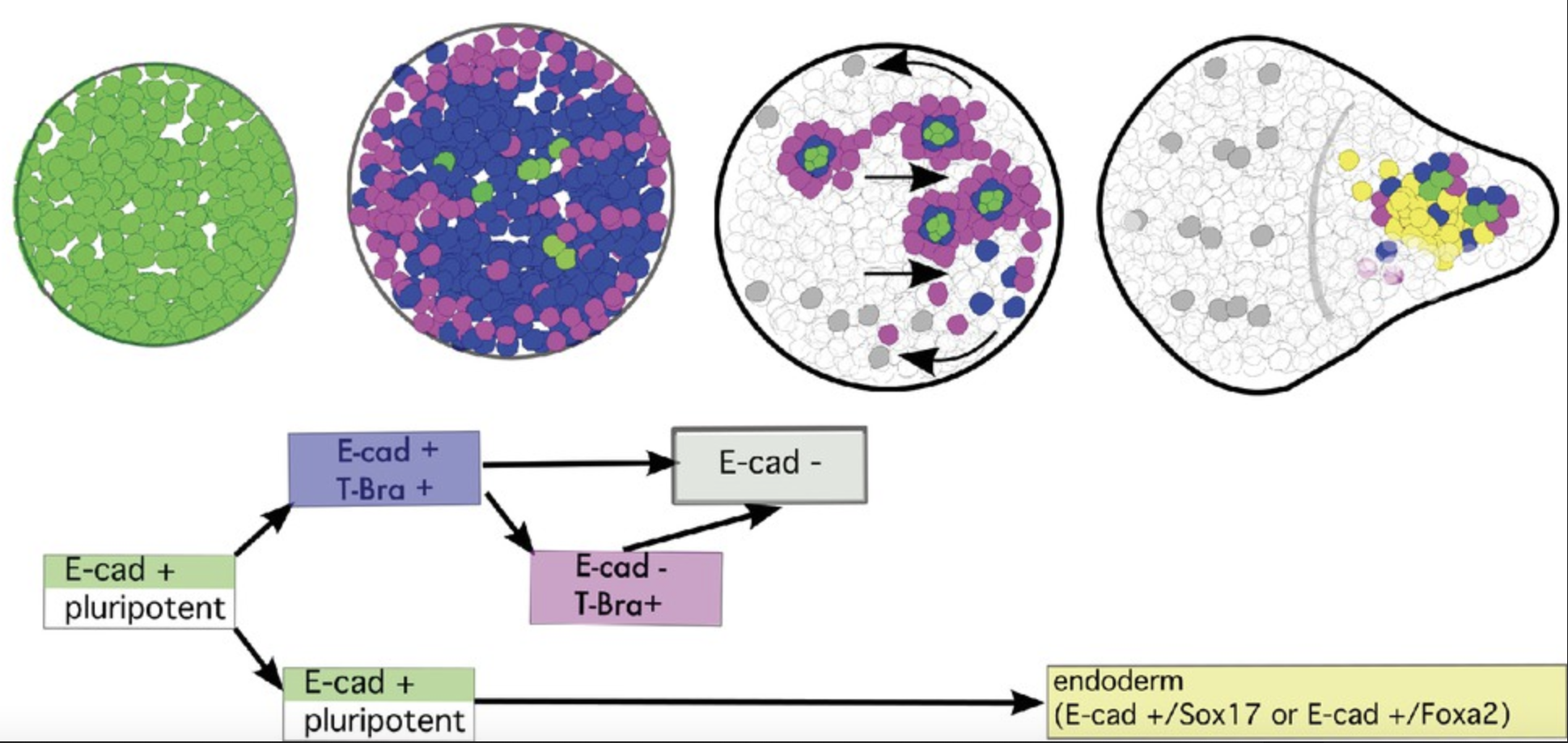
Cell-state transitions and collective cell movement generate an endoderm-like region in gastruloids
The Lenne team published in Elife: using gastruloids –3D aggregates of mouse embryonic stem cells- they study at cellular resolution the specification of the endoderm.
Targeted Tshz3 deletion in corticostriatal circuit components segregates core autistic behaviors
Nos résultats apportent un éclairage nouveau sur l’implication du circuit corticostrié dans les TSA.

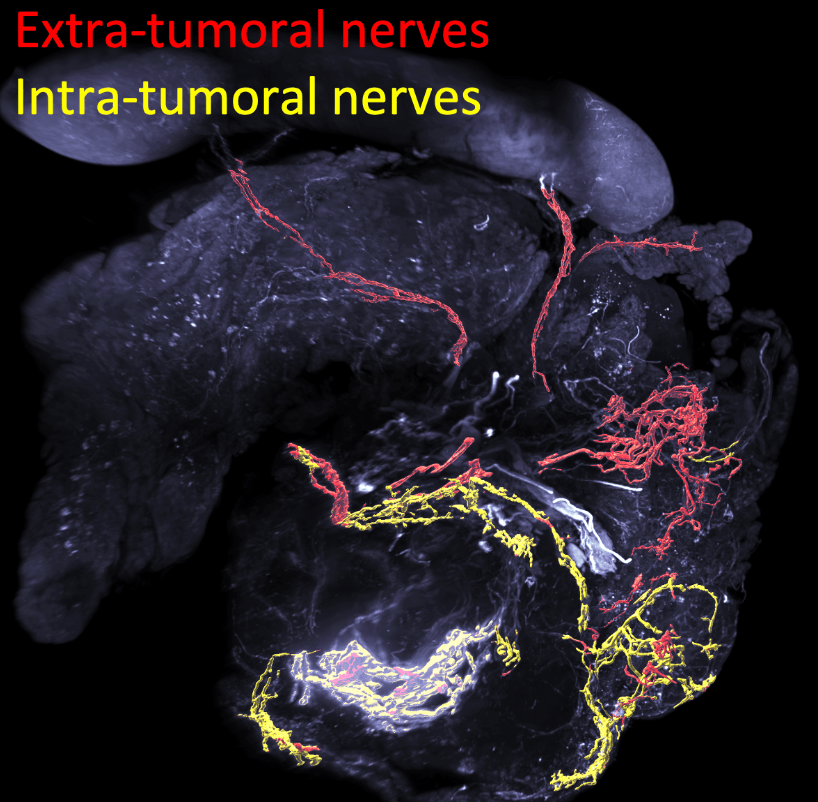
Nervous But Sympathetic !
In this study, the Mann team showed that pancreatic cancer development promotes the sprouting and growth of sympathetic axons.
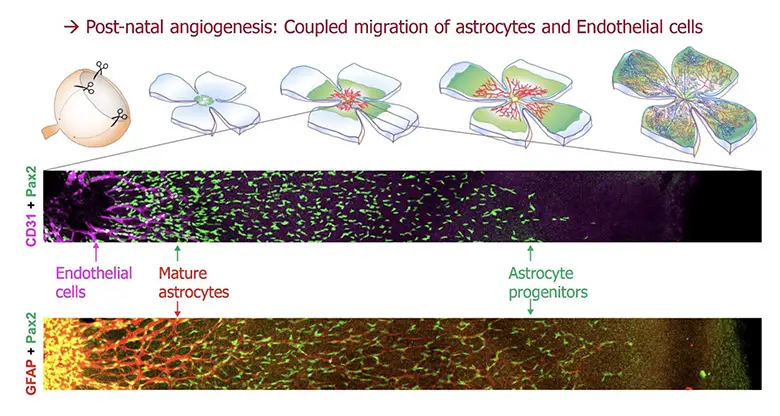
Astrocyte-intrinsic and –extrinsic activities of the Fat1 cadherin regulate astrocyte development and angiogenesis in the mouse retina
The Helmbacher team identified the Fat1 Cadherin as a novel regulator of retinal vascular integrity, and investigated the underlying mechanisms.
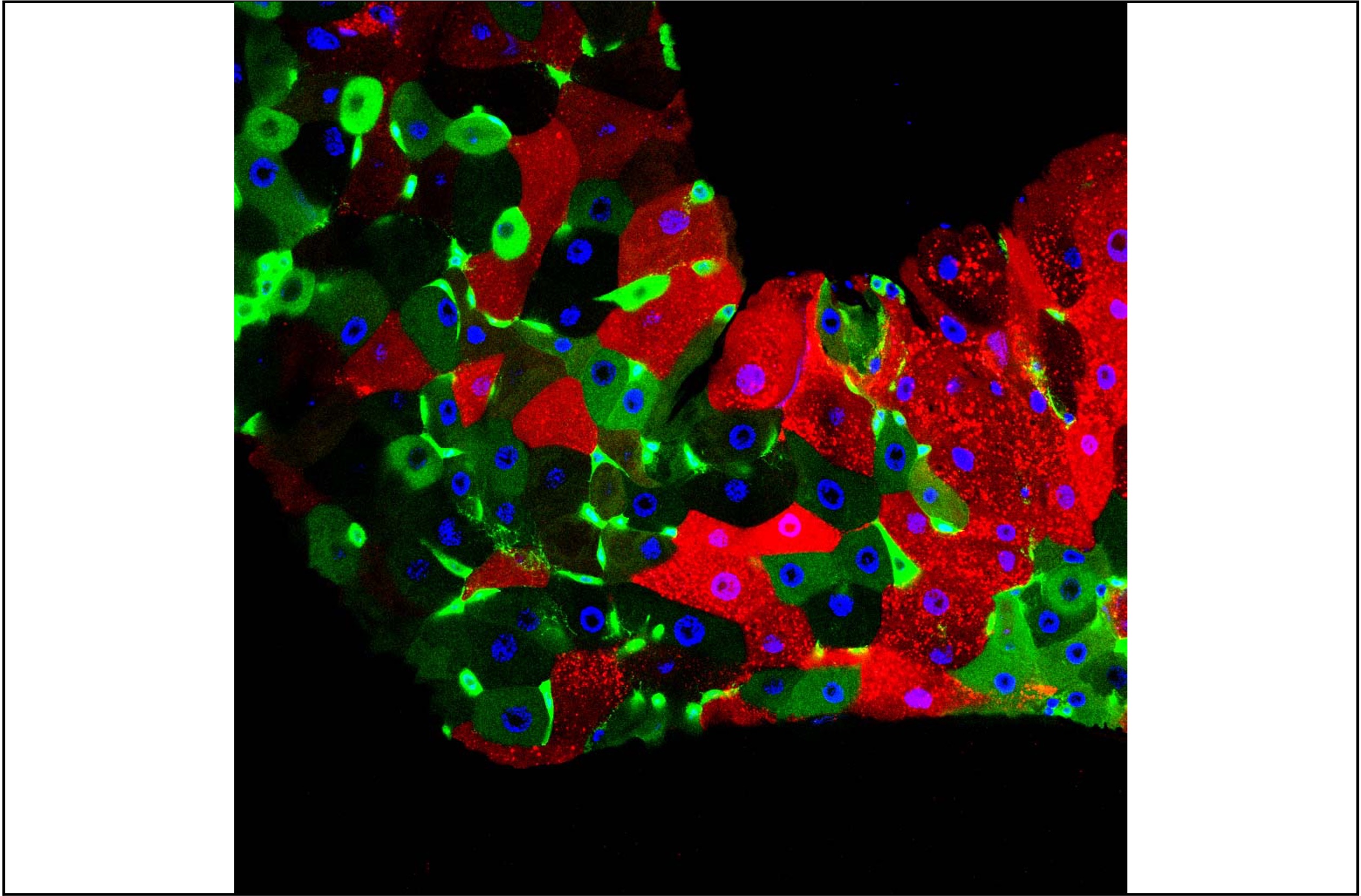
Gut bacteria regulate lipid metabolism in adipocytes
Our work demonstrates that by activating the NF-kB pathway, cell wall peptidoglycan produced by these same bacteria, counteracts SREBP activation in adipocytes in a cell-autonomous manner.
A mechanism to explain the specialization of actin isoforms despite their similarity
Our results allow us to propose a simple and efficient model of how life could generate new functions through the use of new actin isoforms.
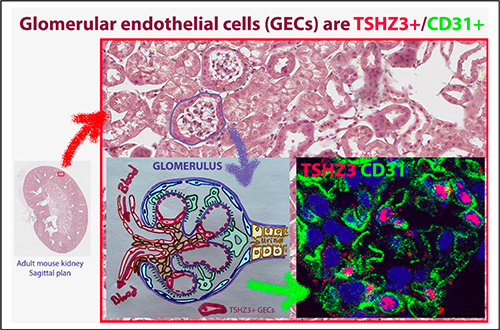
Haploinsufficiency of the mouse Tshz3 gene leads to kidney defects
Data published in Hum Mol Genet. confirm that Tshz3 heterozygous mice constitute a model that replicates many of the clinical problems reported in patients with TSHZ3 heterozygous conditions.
The Marseille Developmental Biology Institute is recruiting group leaders
The selected candidates will receive support to establish a group in a fully renovated building, have access to cutting-edge scientific core facilities, and will be assisted in obtaining a tenured position, and in securing extramural funding.
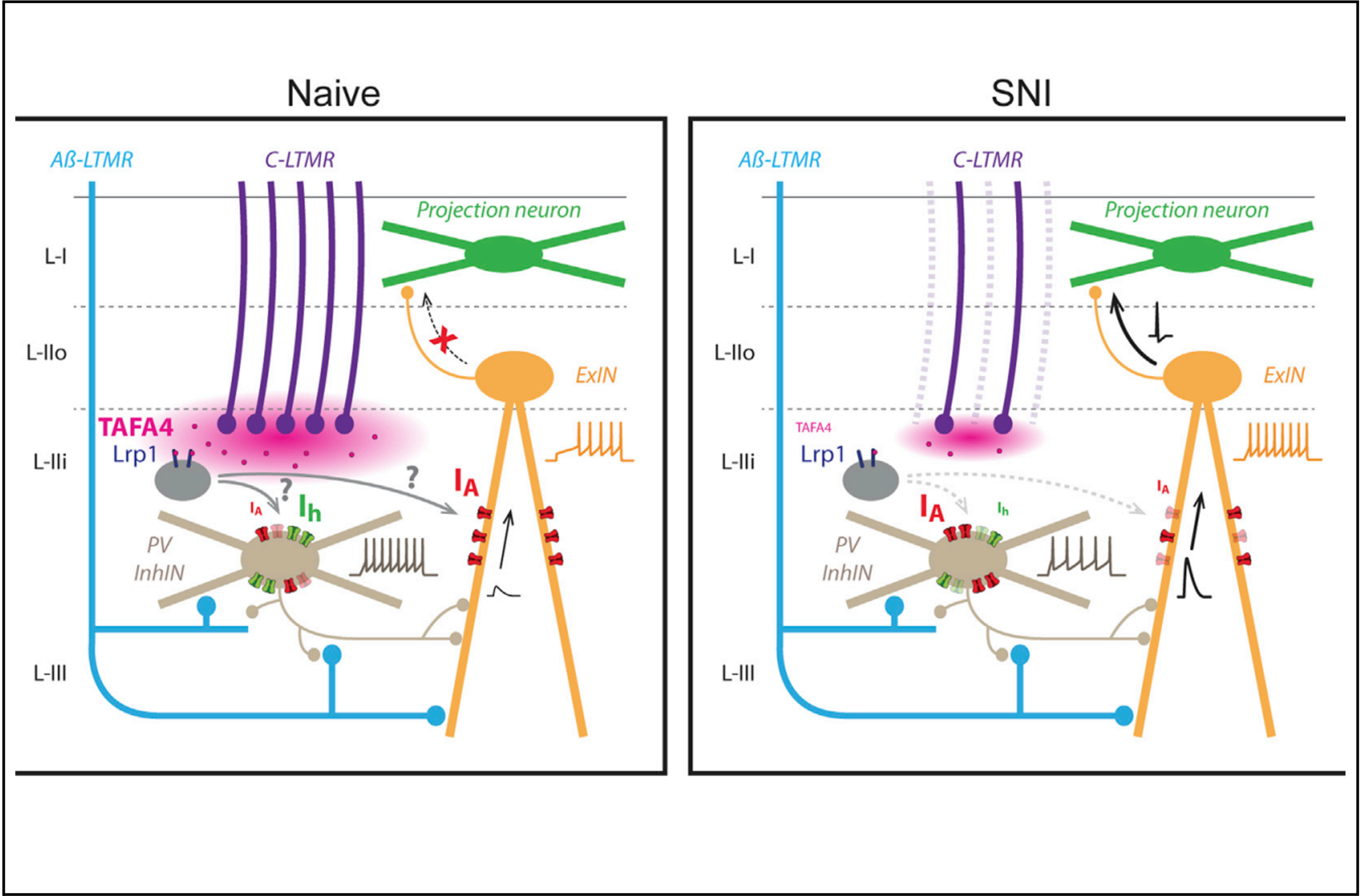
TAFA4 relieves injury-induced mechanical hypersensitivity through LDL receptors and modulation of spinal A-type K+ current
Our findings provide mechanistic insight into the mechanism by which TAFA4 reverses injury-induced mechanical hypersensitivity, by restoring normal spinal neuron activity, and highlight the considerable potential of TAFA4 as a treatment for injury-induced mechanical pain.
Lydia Kerkerian-Le Goff was awarded the Chevalier de la Légion d’Honneur
Lydia Kerkerian-Le Goff, Research Director at the CNRS and head of the Cellular Interactions, Neurodegeneration and Neuroplasticity team at the Marseille Institute of Developmental Biology (IBDM)

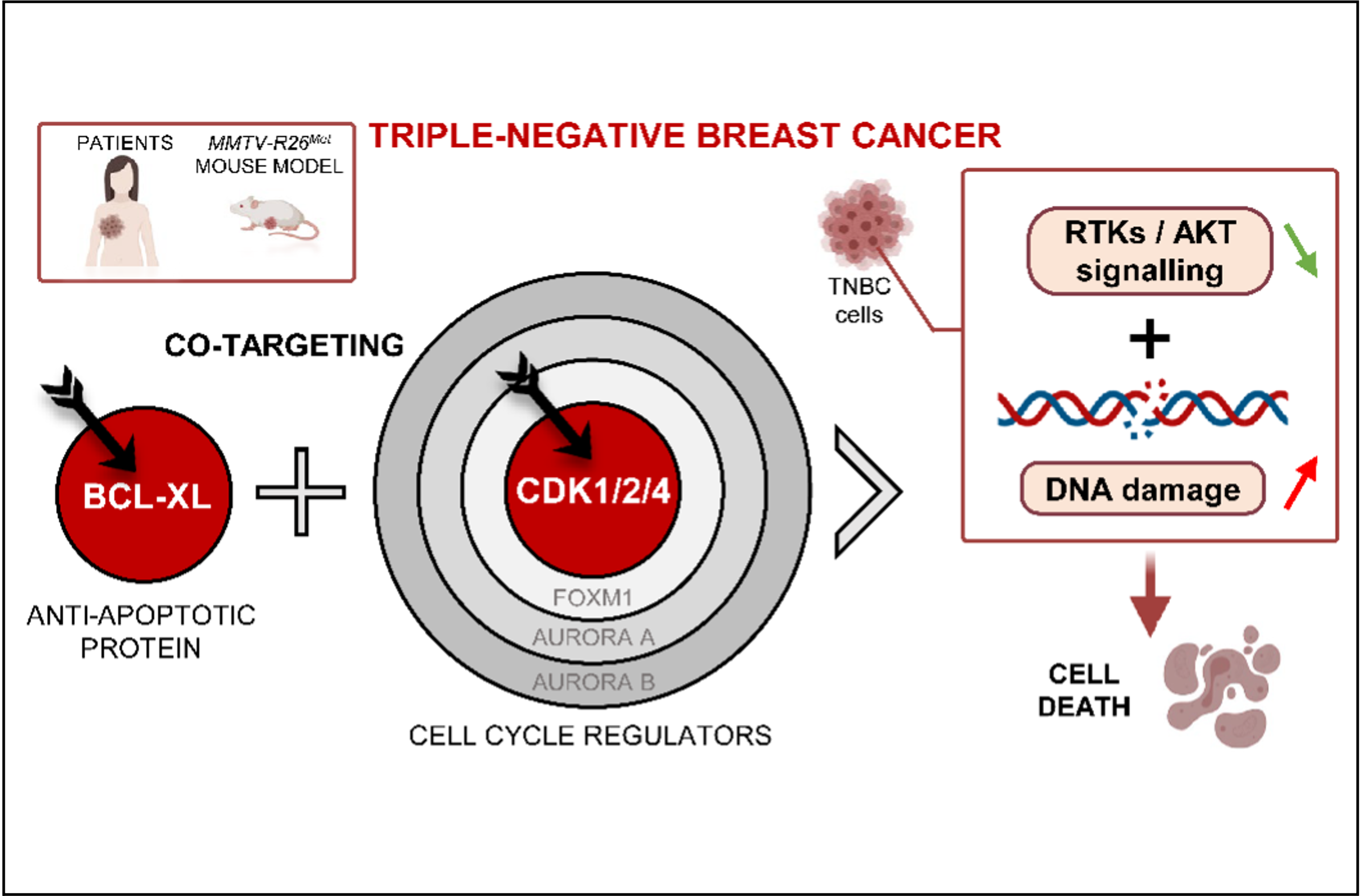
Specificity matters in targeting cell cycle regulators for optimal anticancer response
Through an international collaboration, using breast cancer as a biological paradigm, Flavio Maina Team publishes a work in the Theranostics journal exemplifying how specificity in targeting cell cycle regulators is essential for combinatorial cancer therapies.
Ardem Patapoutian, shares the honours of his Nobel Prize with his former post-docs and students
Ardem Patapoutian shares the honours of his Nobel Prize with his former post-docs and students including Aziz Moqrich, Chronic pain : Molecular and cellular mechanisms

7 IBDM teams have received grants from ANR
…

Formation of polarized contractile interfaces by self-organized Toll-8/Cirl GPCR asymmetry
Jules Lavalou and Qiyan Mao from Thomas Lecuit’s team report that Toll-8 controls myosin-II planar polarity in Drosophila embryos and wing discs via a physical interaction with the GPCR Cirl/latrophilin.

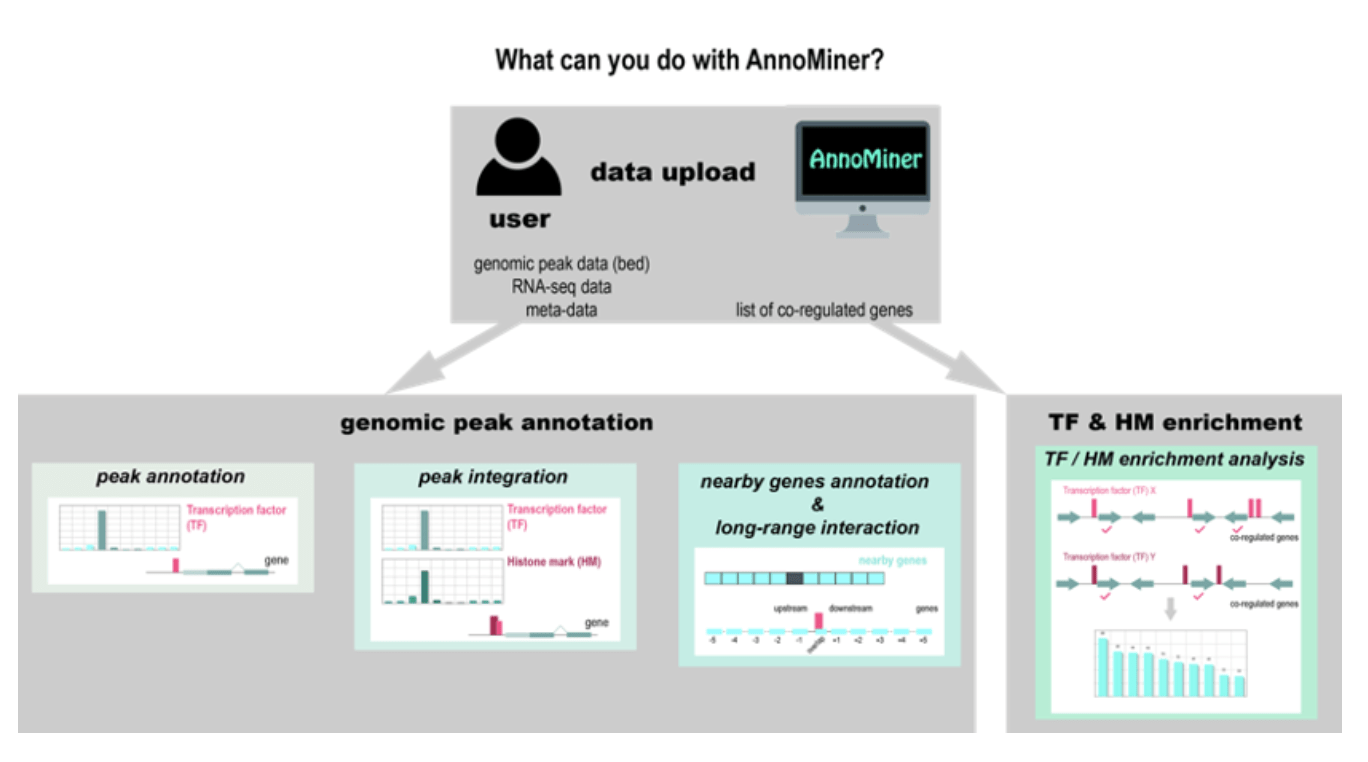
Why not ANNOtate and MINE your ChIP-seq data with AnnoMiner
We introduce a novel, user-friendly web-based tool ‘AnnoMiner’ to annotate and integrate epigenetic and transcription factor binding data.
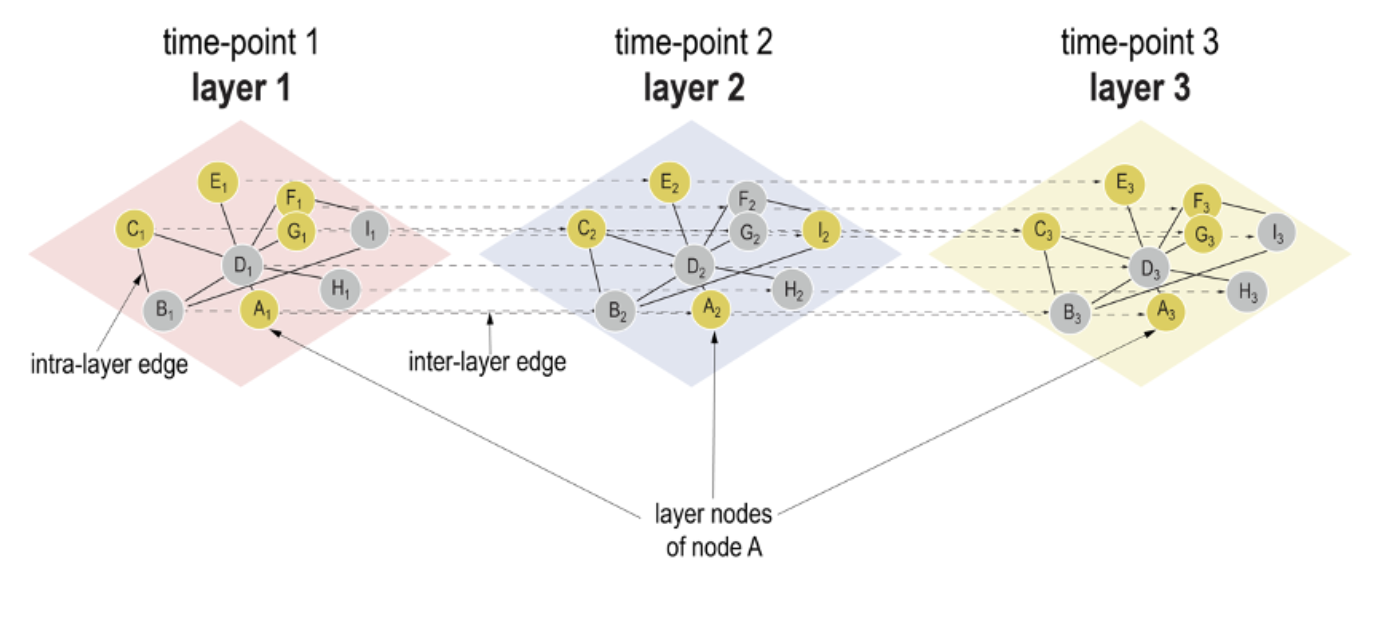
Look at the TIME in your interaction network
The Habermann team has repurposed the concept of multilayer networks generally used to integrate different types of data.
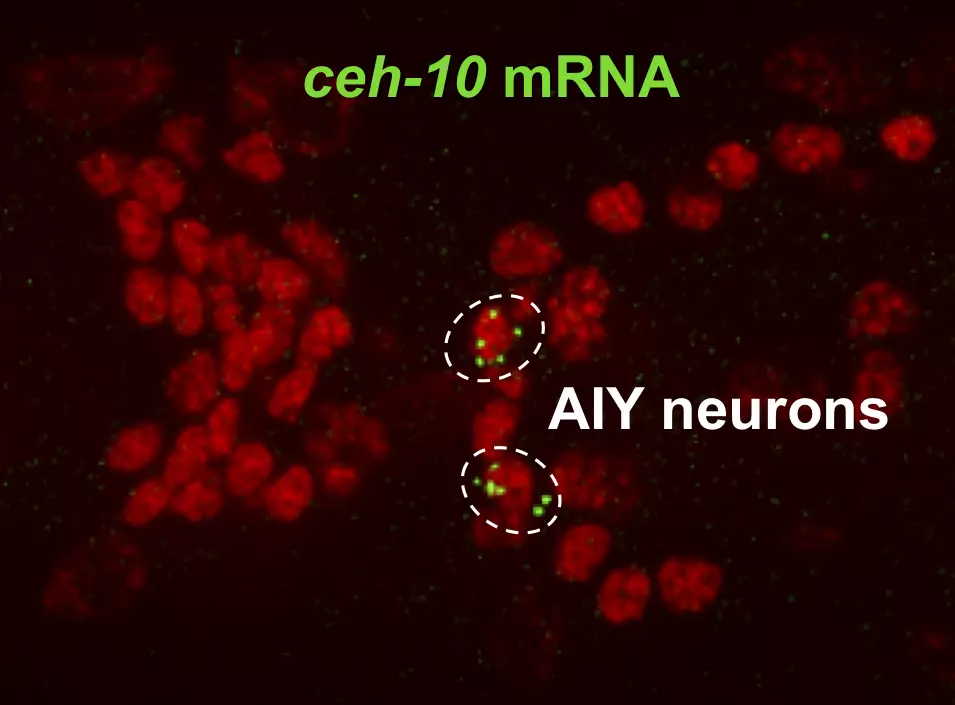
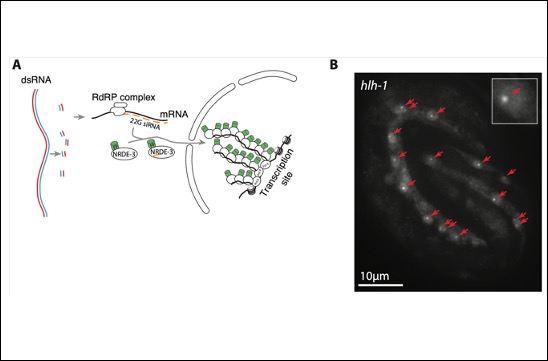
A novel method to image gene expression in vivo
Antoine Barrière and Amel Toudji-Zouaz in the Bertrand team developed a new method to image and quantify transcription in vivo.
TP53INP1, a player in neuron defense against chronic stress
This study published in Cell Death & Disease, conducted by the team of L. Kerkerian-Le Goff provides the first evidence for a role of the stress-induced protein TP53INP1 in the maintenance of neuronal homeostasis in ageing and Parkinson’s disease-related stress conditions.
Tafalgie Therapeutics, a new start-up company, based on the work of Aziz Moqrich’s team
Tafalgie Therapeutics is a new start-up company created in November 2020, based on the work of Aziz Moqrich’s team “Chronic pain: cellular and molecular mechanisms” at IBDM.

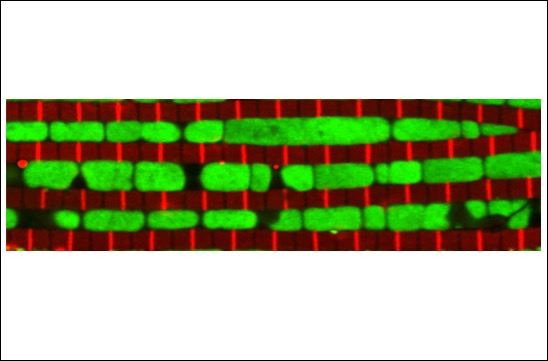
Muscle myofibrils shape their mitochondria and vice versa
By combining genetics in the fruit fly Drosophila with state-of-the-art imaging and deep-learning, researchers at IBDM have found that mitochondria coordinate their formation with myofibril development to match the correct muscle type.
Thesis Prize from the French Society for Developmental Biology (SFBD) for Alexandre CHUYEN
Alexandre CHUYEN, who did his doctoral thesis under the supervision of Andrea Pasini in the Kodjabachian team, was awarded the 2021 Thesis Prize from the SFBD.
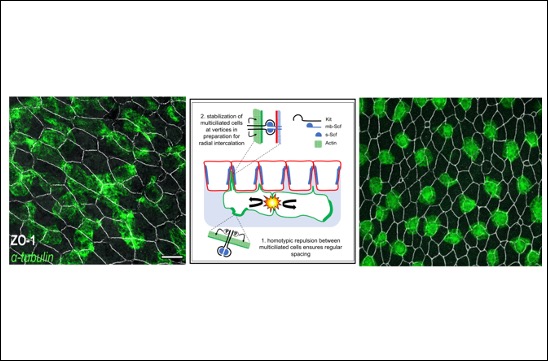
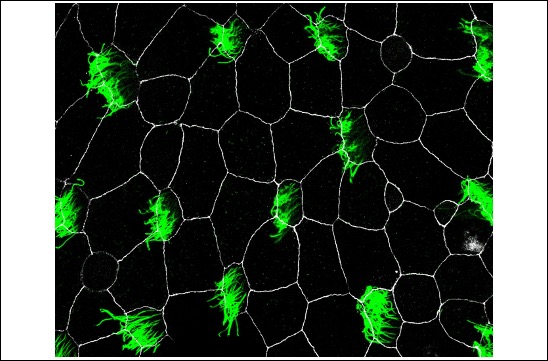
Social distancing : Cells show the way !
A study published in Developmental Cell by the Kodjabachian team reveals how multiciliated cells are distributed at regular intervals in the Xenopus embryonic epidermis.
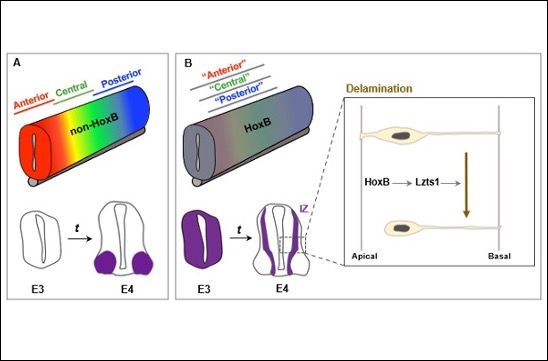
Identification of a generic function for vertebrate Hox proteins in the neural tube
The team of Yacine Graba and Andrew Saurin identified an atypical function for HoxB genes during spinal cord development.
Identification of a family of fluorescent nucleotides binding to actin
The team of Alphée Michelot, in collaboration with the team of Pekka Lappalainen (University of Helsinki) identified and characterized a family of highly sensitive fluorescent nucleotide analogues structurally compatible with actin.
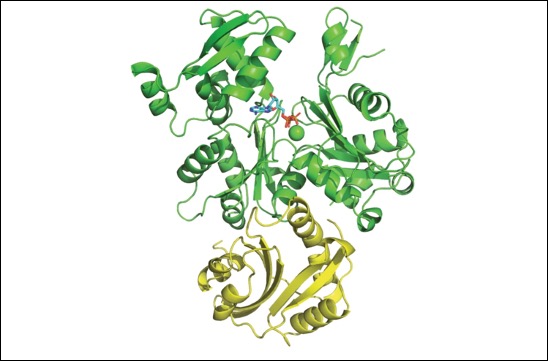
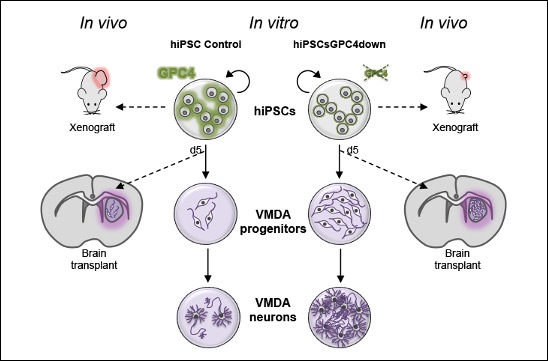
A new target to control the differentiation and tumorigenic potentials of human induced pluripotent stem cells
The teams of F Maina and L Kerkerian-Le Goff publish in Stem Cells Translational Medicine a work led by Rosanna Dono showing that levels of the signal modulator GLYPICAN-4 are critical for the generation of midbrain dopamine neurons from human induced pluripotent stem cells and for overcoming their tumorigenic properties.
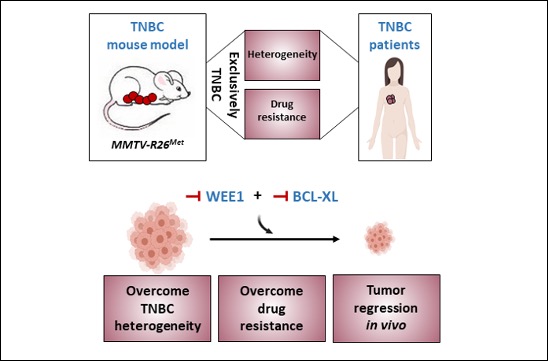
To better understand Triple Negative Breast Cancer in order to develop new therapies
Maina Team publishes in Advanced Science the setup of a genetic system that models, in mice, the heterogeneity and primary resistance to treatment of human Triple Negative Breast Cancer (TNBC).
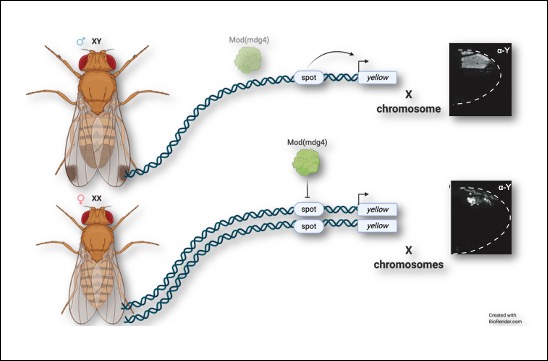
Transvection regulates the sex-biased expression of a fly X-linked gene.
A publication from the Prud’homme team shows how the sexually dimorphic expression pattern of an X-linked gene is controlled by an interallelic interaction, a phenomenon known as transvection.
The Hippo pathway controls muscle growth
Schnorrer team and colleagues discovered that a signalling pathway, called the Hippo pathway, is controlling muscle growth during development of Drosophila flight muscles.
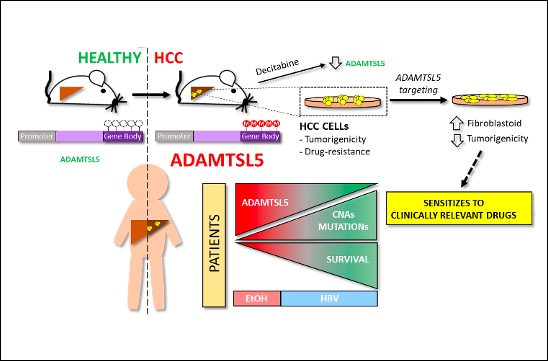
Researchers have identified a new biomarker and potential master regulator of tumorigenesis and drug resistance in Hepatocellular Carcinoma (HCC).
Maina Team publishes in Journal of Hepatology. The team found that ADAMTSL5 is an epigenetically activated oncogene overexpressed in a large fraction of human HCC patients and in HCC mouse models.
EPySeg: a coding-free solution for automated segmentation of epithelia using deep learning
Benoit Aigouy, from the Prud’homme team, developed EPySeg, an open-source software that uses deep learning to segment membrane-stained epithelial tissues, from any organism, automatically and very efficiently.
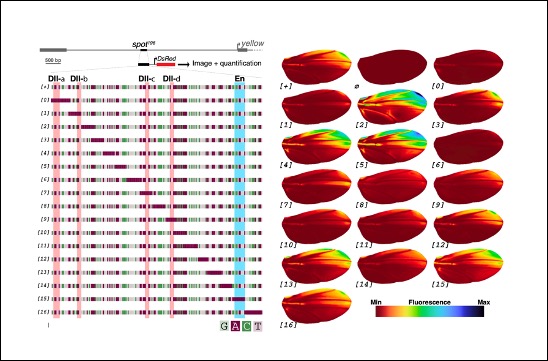
Regulatory encoding of quantitative variation in spatial activity of a Drosophila enhancer
A publication from the Prud’homme team, in collaboration with the group of N. Gompel explores how the spatial pattern and quantitative regulatory information is encoded in the enhancer sequence of a developmental gene.
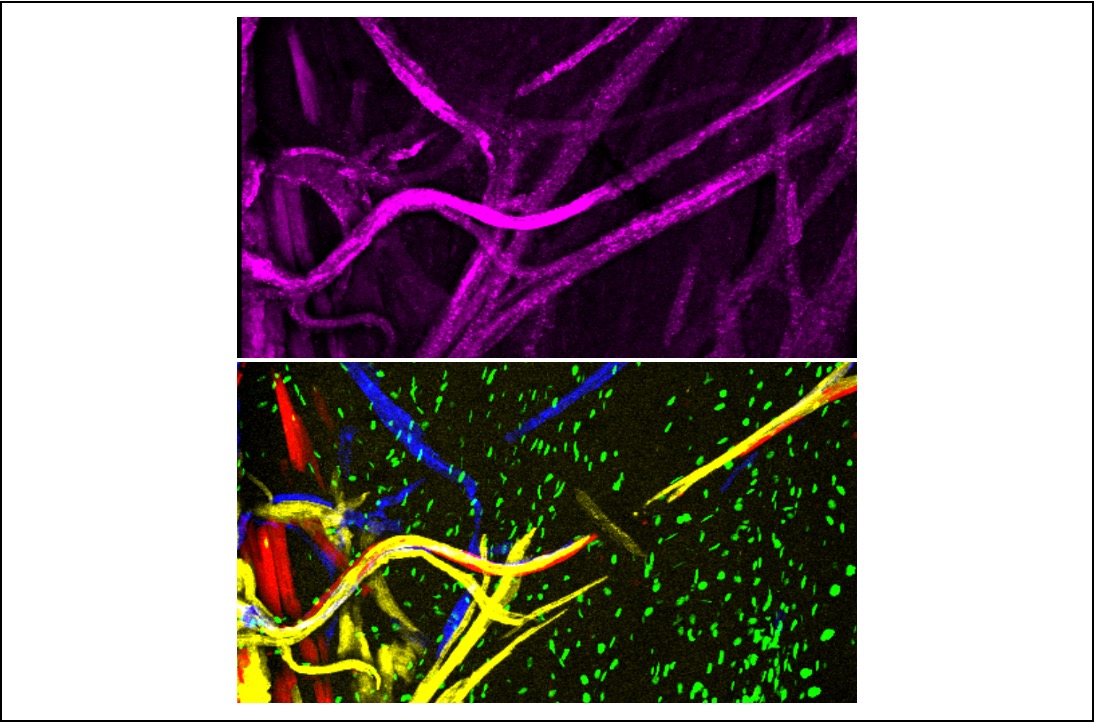
How is the electrical circuit of the mouse heart built?
A publication from the Kelly team in Nature Communications, conducted by Caroline Choquet and Lucile Miquerol, focuses on the development of cardiac Purkinje fibers that serve as electrical cables in the ventricles to synchronize heartbeats.
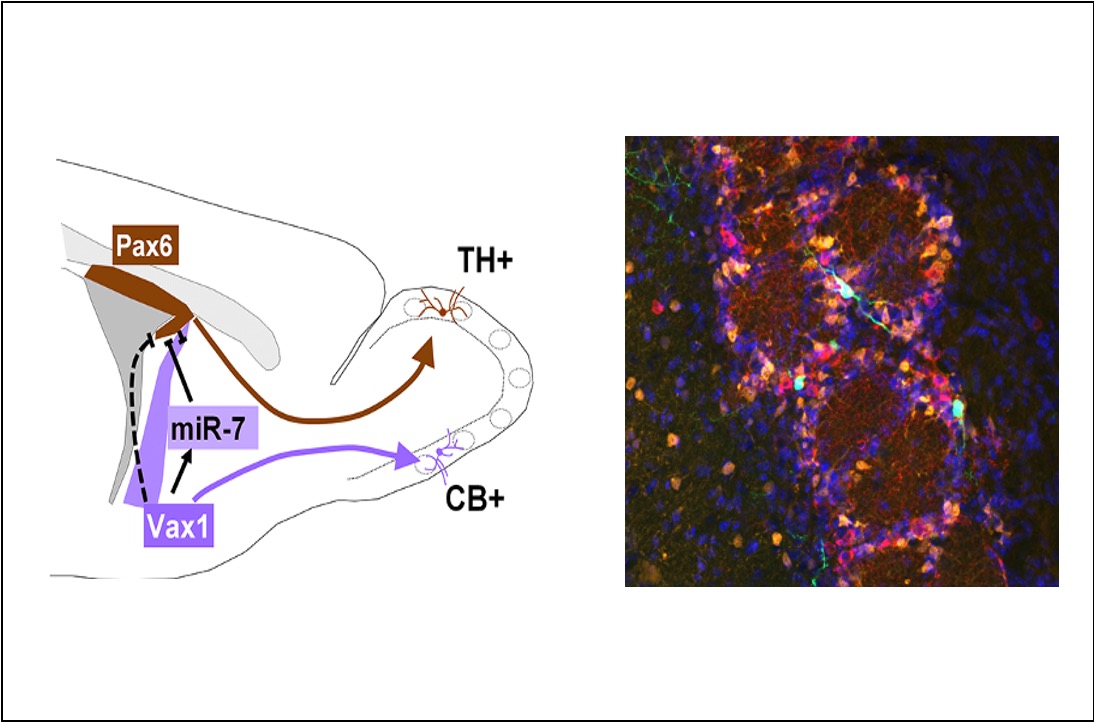
How neural stem cells in the brain are made different
In a paper published in elife, Nathalie Coré from the Cremer team describes a new regulatory cascade, implicating transcription factors and a microRNA, that controls how neural stem cells determine which type of neuron they produce.
Pierre-Francois Lenne elected as EMBO member
Heidelberg, 7 July 2020 – EMBO has bestowed upon 63 leading scientists the lifetime honour of EMBO Membership in recognition of their remarkable achievements in the life sciences, it was announced today.

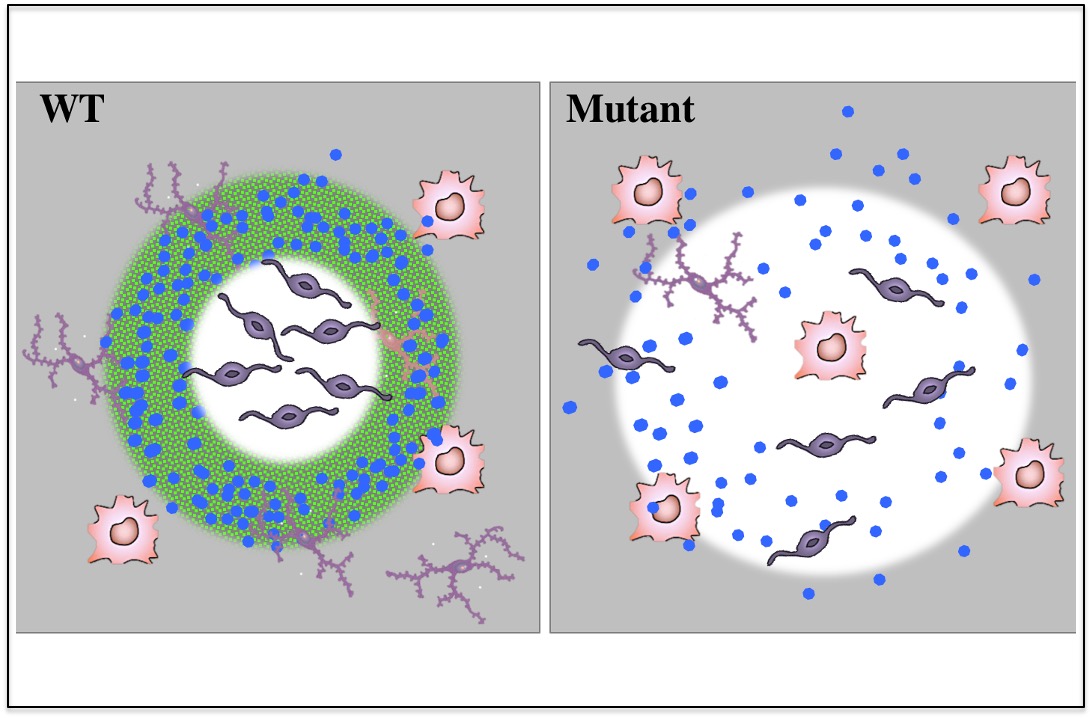
Mature oligodendrocytes bordering lesions limit demyelination and favor myelin repair via heparan sulphate production
A study recently published in elife by the Durbec’s team in collaboration with Anna Williams showed that mature OLG cells spared by the degenerative process surrounding demyelinated lesions also contribute to this repair mechanism by producing HS and modulating Shh signaling pathway.
Drosophila Aversive Behavior toward Erwinia carotovora carotovora Is Mediated by Bitter Neurons and Leukokinin
In a recent article published in iScience, Bernard Charroux, Fabrice Daian and Julien Royet analyze the behavior of fruit flies simply put in contact with bacteria potentially pathogenic to them.
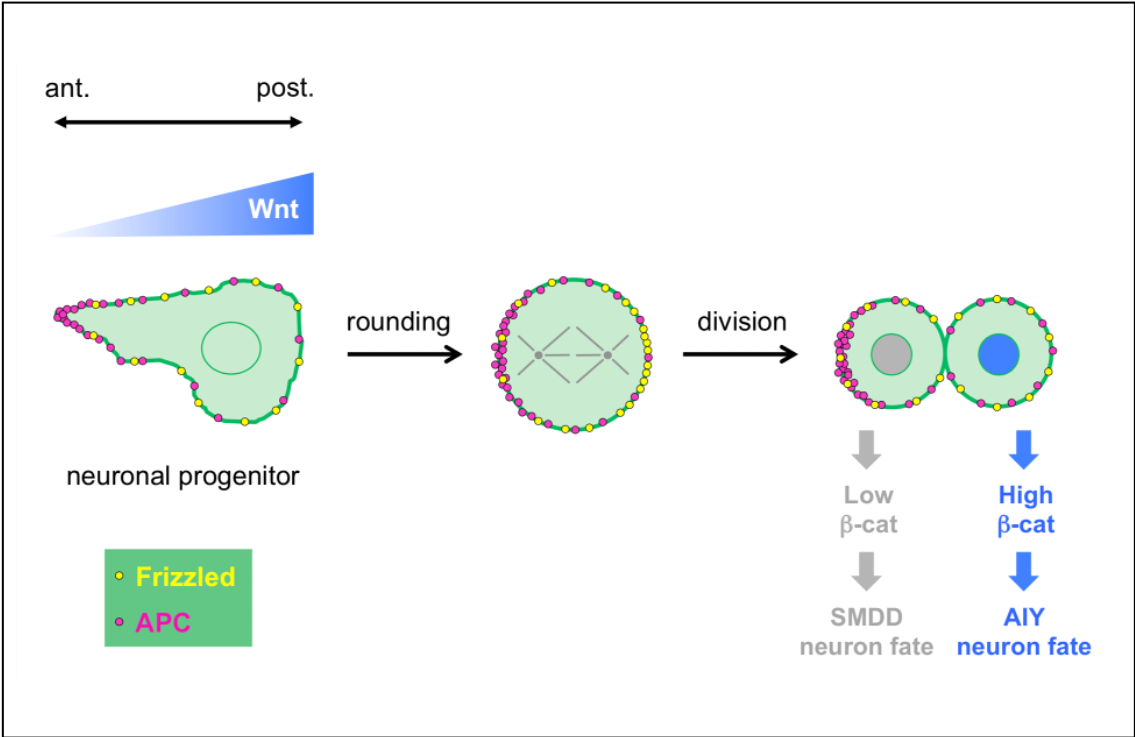
Regulation of neuronal progenitor asymmetric divisions by Wnt ligands
In a collaborative study published in Development, the teams of Vincent Bertrand and Pierre-François Lenne analyze the role played by Wnt ligands in the divisions that generate neurons during nervous system development.
Cardiopharyngeal mesoderm origins of musculosketelal and connective tissues in the mammalian pharynx
In a study published in Development, Adachi et al used genetic lineage tracing to investigate the cellular origin of the throat in the mouse model.
Loss of bhlha9 impairs thermotaxis and formalin-evoked pain in a sexually dimorphic manner
In their study published in Cell Reports, Bohic et al. confirm the role of C-LTMRs in inflammatory pain modulation by inactivating the transcription factor bhlha9 particularly enriched in these touch neurons.
Maina Team in collaboration with the imXgam research team of CPPM and researchers at the CIML and Cerimed, publishes in iScience
Researchers have used the CERN pixels to follow tumour evolution in a liver cancer mouse model.
Mechanical stiffness of reconstituted actin patches correlates tightly with endocytosis efficiency
A multi-disciplinary collaboration between the team of Alphée Michelot and the team of Olivia du Roure and Julien Heuvingh at the ESPCI publishes in Plos Biology a study demonstrating that the mechanical stiffness of endocytic actin patches correlates tightly with endocytosis efficiency.
Neuronal integration in the adult mouse olfactory bulb is a non-selective addition process
A recent study conducted by Jean-Claude Platel in Harold Cremer’s team published in eLife showed that these newly generated neurons add up permanently in the olfactory bulb neural network leading to an increase in the neuronal population and the size of the olfactory bulb.
Mechanisms of brain cancers with early developmental origins revealed in Drosophila
In a recent work published in eLife, Cédric Maurange’s team identifies a genetic program that organizes the metabolic heterogeneity of cells in a pediatric neural tumor model.
Peptidoglycan-dependent NF-κB activation in a small subset of brain octopaminergic neurons controls female oviposition
Julien Royet’s team uses the genetic tools generated in the fruit fly to dissect the molecular interactions between bacteria and the host’s nervous system.
European Synergy Grant for Muscle Research
The European Research Council (ERC) awarded one of the rare ERC Synergy Grants to an international consortium of scientists, Frank Schnorrer, Stefan Raunser, Dirk Görlich and Mathias Gautel.
Shaping Life 2
…
Embryo development like a stadium wave
In a recent study appeared on the international journal Nature, Thomas Lecuit and his colleagues at the Institut de Biologie du Développement de Marseille describe how tissue shape changes are self-organized.
Size regulation of actin networks in cells
The team of Alphée Michelot publishes in Plos Biology a study demonstrating how cells control the size of multiple actin networks within a common cytoplasm.
