Team members
Stem cells and brain repair
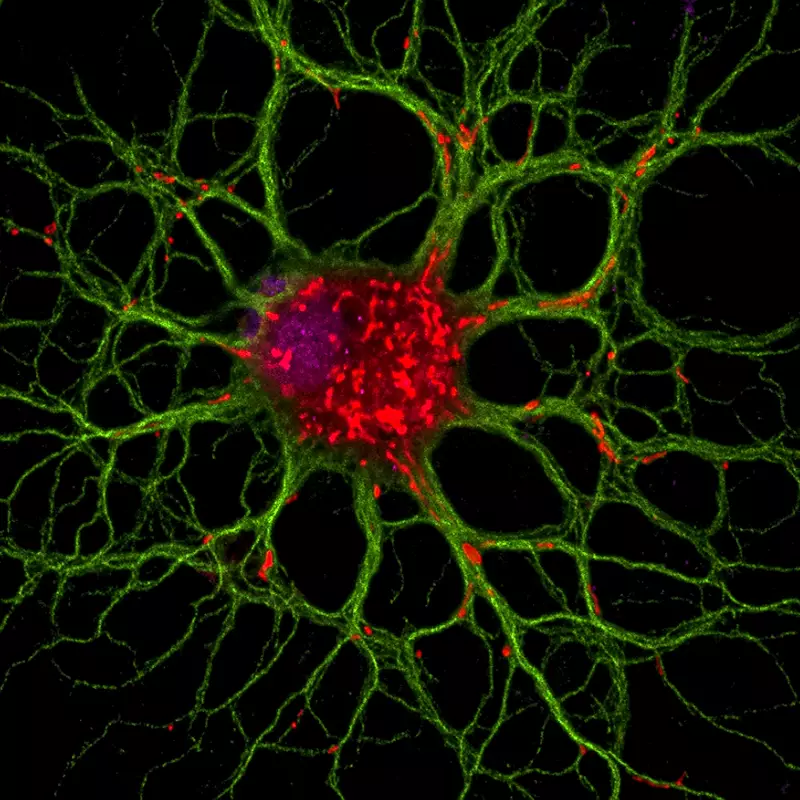
Our team seeks to understand the repair process in the adult brain and more specifically the myelin regeneration.
Myelin is a sheath that surrounds and insulates the nerve, and improves signal transmission. When myelin is damaged or abnormal, due to genetic abnormalities, trauma or neurodegenerative diseases such as multiple sclerosis, neurological disorders occur resulting in severe disability.
The nervous system is capable, to a certain extent, to regenerate the myelin sheath, but this self-repair process is incomplete and insufficient. This regeneration is possible thanks to the presence in the brain of stem cells and progenitor cells that can replace oligodendrocytes the myelin forming cell. It is therefore important to understand all the mechanisms involved in the process of myelin repair in order to develop new therapeutic strategies.
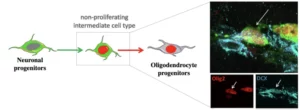
Our research aims to better understand the biology of adult progenitor and stem cells involved in remyelination. Our goal is to decipher the cellular and molecular mechanisms controlling the mobilization of these cells. We are trying to determine which cells are involved in the repair process. How these cells migrate to the lesion site? What are the factors that control their differentiation into oligodendrocytes in physiological and pathological conditions? We are looking for factors that can influence or control these events when administered to the animal. We use the mouse as a model organism and combine techniques of cell biology, molecular imaging and neurosurgery to answer these questions.
Publications
Our last publications
Top
All
Top
Region and dynamic specificities of adult neural stem cells and oligodendrocyte precursors in myelin regeneration in the mouse brain
B.Brousse
Et Al.
K.Magalon
P.Durbec
M.Cayre
All
Region and dynamic specificities of adult neural stem cells and oligodendrocyte precursors in myelin regeneration in the mouse brain
B.Brousse
Et Al.
K.Magalon
P.Durbec
M.Cayre
Oligodendrogenesis in the normal and pathological central nervous system.
E.Waly B
Et Al.
M.M
C.M
D.P.
RAE-1 is expressed in the adult subventricular zone and controls cell proliferation of neurospheres.
Control of planar divisions by the G-protein regulator LGN maintains progenitors in the chick neuroepithelium.
M.X
Et Al.
J.F
�.P.
News
of the team
Articles
Job opportunities
Articles
3 motivated and talented students successfully defended their thesis between February and July 2023.
5 motivated and talented students successfully defended their thesis between September 2022 and January 2023.
10/08/2021
7 IBDM teams have received grants from ANR
7 IBDM teams have received grants from the Agence Nationale pour la Recherche (ANR) in 2021. Congratulations to Vincent Bertrand, Harold Cremer, Pascale Durbec, André Le Bivic, Pierre-François
A study recently published in elife by the Durbec’s team in collaboration with Anna Williams showed that mature OLG cells spared by the degenerative process surrounding demyelinated lesions also contribute to this repair mechanism by producing HS and modulating Shh signaling pathway.
Job opportunities
No jobs opportunities found..
Lab members
They drive our research
Researcher
Alumni
They contributed to our research
Magali Macchi
Enseignant chercheur chez Aix-Marseille Université
Xavier Morin
CNRS Researcher
Françoise Padilla
Responsable Equipe Imagerie Photonique
Fabienne Alfonsi
Medical Marketing Director chez Teoxane Laboratories